

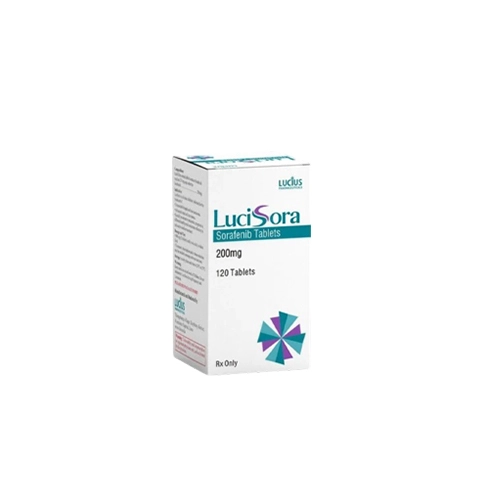
LuciSora (Sorafenib 200mg) Rx
Ask For Price
Lucius pharma
Generic Sorafenib
Pack Size: 120’s Pot
Form: Tablet
Sorafenib, (LuciSora) works for treating Hepatocellular Carcinoma (HCC), Differentiated thyroid carcinoma (DTC), Advanced Renal cell carcinoma (RCC). Sorafenib is a doctor prescription (Sorafenib) medicine.
Lucius Pharma Ltd. in Laos mfg LuciSora. Take this medicine in the dose and duration as advised by your doctor. Swallow it as a whole. Do not chew, crush or break it. LuciSora tablet may be taken with or without food, but it is better to take it at a fixed time.
Suggestion:
Genuine medicine is important for treatment. So, always try to source genuine medicines from authentic sources and check the quality of medicines.
Side effects:
The most common side effects of Sorafenib are:
mouth sores, hair loss, voice changes, or tiredness, weight loss, nausea/vomiting, diarrhea, loss of appetite, changes in taste, dry skin. Tell your doctor or pharmacist right away if any of these side effects persist or worsen.
Remember that your doctor has prescribed this medication because your doctor has judged that the benefit to you is greater than the risk of side effects. Many people using this medication do not have serious side effects.
Tell your doctor right away if you have any serious side effects, including: easy bruising/bleeding, signs of liver disease (such as nausea/vomiting that doesn’t stop, stomach/abdominal pain, yellowing eyes/skin, dark urine), numb/tingling skin, slow wound healing, muscle spasms, signs of infection (such as sore throat that doesn’t go away, fever), signs of heart failure (such as shortness of breath, swelling ankles/feet, unusual tiredness).
Take this medication regularly to get the most benefit from it. To help you remember, take it at the same times on the days your doctor directs.
This is not a complete list of possible side effects. Contact your doctor or pharmacist if you have any other side effects not listed above.
















Reviews
There are no reviews yet.