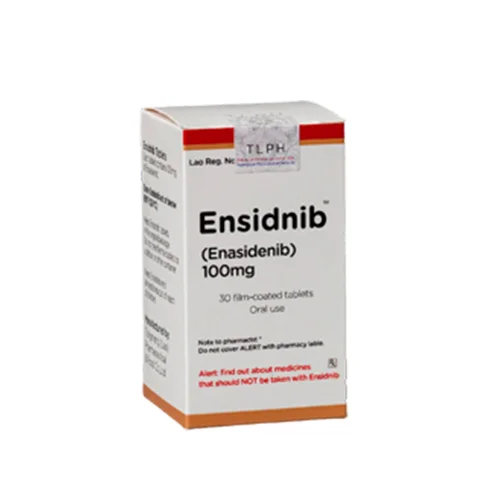
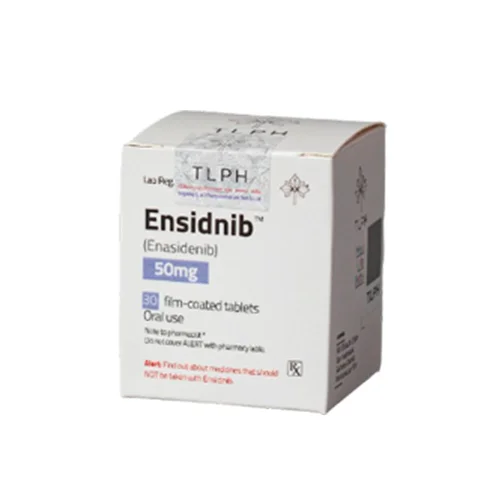
Ensidnib (Enasidenib 100mg / 50mg) Rx
$149.00 – $210.00Price range: $149.00 through $210.00
TLPH pharma
Generic ENASIDENIB
Pack Size: 30’s Pot
Form: Tablet
ENASIDENIB, (Ensidnib) works for treating patients with acute myeloid leukemia (AML) with an isocitrate dehydrogenase-2 (IDH2) mutation whose disease has come back or has not improved after previous treatments. Enasidenib helps the bone marrow produce healthy blood cells and reduces the need for blood transfusions. ENASIDENIB a doctor prescription (ENASIDENIB) medicine.
TLPH Pharma Ltd. In Laos mfg Ensidnib.
Take this medicine in the dose and duration as advised by your doctor. Swallow it as a whole. Do not chew, crush or break it. Ensidnib tablet Tablet may be taken with or without food, but it is better to take it at a fixed time.
Suggestion:
Genuine medicine is important for treatment. So, always try to source genuine medicines from authentic sources and check the quality of medicines.
Side effects:
The most common side effects of ENASIDENIB are:
diarrhea, vomiting, Nausea, Nausea and vomiting, or loss of appetite may occur.
Tell your doctor or pharmacist right away if any of these side effects persist or worsen.
Remember that your doctor has prescribed this medication because your doctor has judged that the benefit to you is greater than the risk of side effects. Many people using this medication do not have serious side effects.
Take this medication regularly to get the most benefit from it. To help you remember, take it at the same time each day.
This is not a complete list of possible side effects. Contact your doctor or pharmacist if you have any other side effects not listed above.
| Strength |
100mg ,50mg |
|---|---|
| Pack Size |
30's Pot |

















Reviews
There are no reviews yet.